Insect Collecting 101
The key to getting different insects is to collect in different habitats at different times of the day. Remember some insects only come out at night.
Places to collect include:
- Your own yard. In particular look for plants with lots of aphids or other pests, as these plants may also have predatory insects.
- A grassy field.
- Near lights at night. Lighted gas stations in "the middle of nowhere" are usually a goldmine of insects.
- In decaying logs, under stones, beneath soil and plant debris.
- Some important equipment for collecting include: insect net, killing jar, forceps, relaxing jar, spreading board, pinning block, insect pins and labels, and a storage box.
- Click here for Texas A&M University Resources: Insect Collecting and Classroom Use
- By hand. Be careful of biting or stinging insects!
- Collecting Techniques: Bee Observant video (swf 3.5MB) - Rebecca Baldwin, UF/IFAS

- Collecting Techniques: Bee Patient video (swf 3.0MB) - Rebecca Baldwin, UF/IFAS

- Collecting Techniques: Bee Cautious video (swf 4.1MB) - Rebecca Baldwin, UF/IFAS

- Collecting Techniques: Bee Observant video (swf 3.5MB) - Rebecca Baldwin, UF/IFAS
- Plastic bags. Put the open bag over the insect to catch it, and place the bag in the freezer.
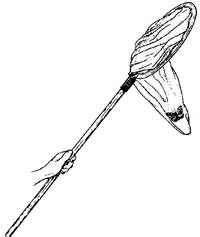
- Insect net. You can purchase one at supply companies or even local department stores. Or you can make your own out of nylon mosquito netting (available at a army surplus store) or any other netlike material. If you would like to make your own net, there are directions at the back of the packet. To use an insect net, sweep it back and forth several times as you walk through foliage.
- Collecting Techniques: Sweep Nets video (swf 3.2MB) - Rebecca Baldwin, UF/IFAS

- Collecting Techniques: Sweep Nets video (swf 3.2MB) - Rebecca Baldwin, UF/IFAS
- Beat sheet. Simply lay a white sheet beneath a tree or bush and hit the leaves of the plant with a stick or broom several times so that the insects fall onto the white sheet below.
- Remove bark from dead or living trees.
- Collecting Techniques: Barking (swf 4.7MB) - Rebecca Baldwin, UF/IFAS

- Collecting Techniques: Barking (swf 4.7MB) - Rebecca Baldwin, UF/IFAS
- Various traps:
- Collecting Techniques: Pitfall Traps video (swf 5MB) - Rebecca Baldwin, UF/IFAS

- Collecting Techniques: Pitfall Traps video (swf 5MB) - Rebecca Baldwin, UF/IFAS
- Freeze them. Place collected insects inside small Tupperware containers or plastic bags and put them in the freezer for a few hours to kill them. Insects can remain in the freezer for several weeks until you are ready to pin them. The insects should be removed an hour before pinning, so they have time to thaw and become soft and movable.
- Use a kill jar. This is a special glass jar with Plaster of Paris on the bottom that you saturate with nail polish remover or some other poison. These can be purchased at biological supply company or constructed.
- Insect pins may be obtained through your county agent or one of the biological supply houses. Common straight pins should NOT be used because they will rust and ruin the specimen.
 Insect pins are available in several sizes; however, sizes No. 2 or 3 are most commonly used.
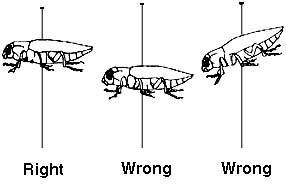
Insect pins are available in several sizes; however, sizes No. 2 or 3 are most commonly used. - An insect is large enough to pin if you can put the pin through the designated area without breaking or distoring it. The pin should always be inserted through a definite part of the body from top to bottom. Be sure the insect is on the pin straight from all viewing angles.
- In the picture to the right, the dots show where to put the pin for different types of
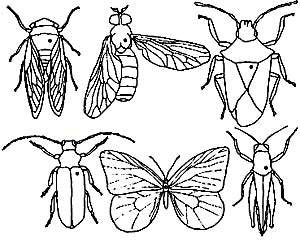 insects. The pin is inserted into the top of the insect's body and pushed through most of the way, leaving enough of the pin at the top to hold the pin without handling the insect itself. The pin must go through far enough that the specimen can be pinned to a piece of Styrofoam without the legs touching the surface. After being pinned and before being allowed to dry, the legs, wings, and antennae should be arranged so they are suitable to study.
insects. The pin is inserted into the top of the insect's body and pushed through most of the way, leaving enough of the pin at the top to hold the pin without handling the insect itself. The pin must go through far enough that the specimen can be pinned to a piece of Styrofoam without the legs touching the surface. After being pinned and before being allowed to dry, the legs, wings, and antennae should be arranged so they are suitable to study.
NOTE: Some long thin insects such as praying mantids or walking sticks may need pieces of thick paper or cardboard beneath the insect to help support it.
- The height of the specimens on the pin should be uniform and a pinning block is used to measure this accurately. Pinning blocks can be purchased from biological supply companies or constructed.
- You should pin an insect quickly after it dies or within a few hours of removing it from the freezer since it will be "soft", and there is less likelihood of the legs and antennae breaking off. You can use other pins to construct a support system to hold legs and antennae in particular positions. After 2 or 3 days your insect will "harden" and remain permanently in this position. A relaxing jar can help if your insect dries out.
- Butterflies, moths, and often grasshoppers, dragonflies, flies, and wasps should have the wings on one or boths sides spread. A spreading board is necessary for this. These can be purchased at a biological supply company or easily constructed. For butterflies, you will want to spread the wings of the butterfly while it is still pliable and use strips of paper to hold the wings in place; after a few days the insect will dry in this position.
- Making a Spreading Board for Pinning Insect Wings – Purdue University Extension
- Lepidoptera Spreading video (swf 24.8MB) - Rebecca Baldwin, UF/IFAS

- Place pinned insects on pieces of Styrofoam.
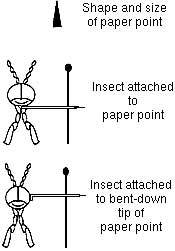
- Small insects (too small to stick a pin through) are "pointed" on small pieces of stiff paper. Cut small triangular pieces of index cards (or other thick white paper), and stick the pin through the piece of paper. Place a small dab of glue on
- the paper and glue it to the right side of the insect. PRACTICE MAKES PERFECT; this is very hard to do. You may have to try 5 or 10 times before perfecting the technique.
- Soft-bodied insects, such as termites, lice, aphids, thrips, etc., should be placed in 70% alcohol (rubbing alcohol) in glass vials with a rubber stopper or plastic cap. If you are using a rubber stopper, the vial needs to be "burped" to release the pressure. This can be accomplished by placing a safety pin or paper clip between the stopper and vial and release the air. A hissing sound will occur. If this is not done, then the stopper will pop out due to the pressure.
NOTE: Sometimes ants can go after your insect collection and eat it. If your collection is ant-infested, put it in the freezer for a few hours to kill the ants. Try to keep your collection in an ant proof box or container to prevent problems.
- During the course of preparation and mounting, insect should always have some type of temporary label or number gining its date and place of collection. Before insect are put away, they should be labeled with a permanent label. It is recommended to print on slightly thicker paper.
- The two types of labels are Date and Locality Label and Identification Label.

- Date and Locality Label: The place of collection should be given as accurately as possible so that it can be found on a good map. The day, month (abbreviation or roman numeral), and year should be given. The collector should be given (this should be you).

- Identification Label: The order and common name (or scientific family name) of the insect should be on this label.
- Date and Locality Label: The place of collection should be given as accurately as possible so that it can be found on a good map. The day, month (abbreviation or roman numeral), and year should be given. The collector should be given (this should be you).
- For pinned and pointed insects, the font size on both of these labels should be 4pt Arial font and the edges of the labels should be cut down to minimize the amount of white boarder around
 the text. They should also be of uniform size. Printing these labels on a Laser printer is highly suggested as they will be much easier to read at the 4pt. size. Labels should be in line with insect. Click here for an example of pin labels that you can type over with your information.
the text. They should also be of uniform size. Printing these labels on a Laser printer is highly suggested as they will be much easier to read at the 4pt. size. Labels should be in line with insect. Click here for an example of pin labels that you can type over with your information. - The three steps of your pinning block should correlate with:
Insect (highest), Date and Locality Label (middle step), Identification Label (lowest step.) - If the specimen is in a vial of alcohol, the font size on both of these labels should be 8pt Arial font. The two labels need to be placed back to back (with the text of each facing outward) and placed inside the vial. Note: DO NOT place Ink Jet printed labels inside the vial of alcohol as many inks dissolve in the alcohol. These labels need to be printed on a Laser printer. Click here for an example of vial labels that you can type over with your information.