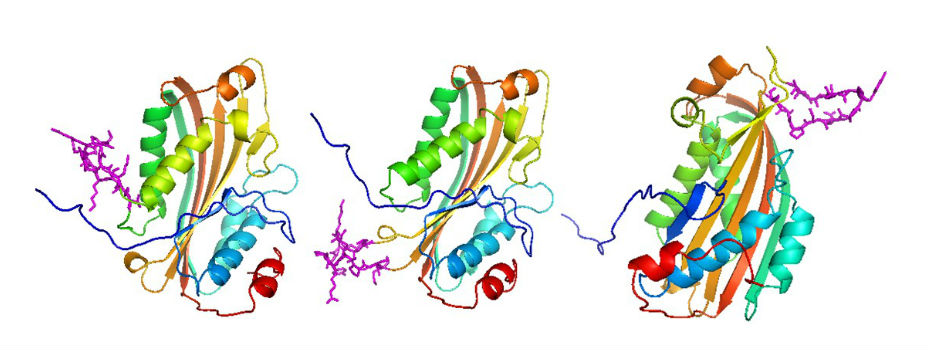
Bt Pesticidal Proteins
Insect specific pesticidal proteins derived from bacteria such as Bacillus thuringiensis (Bt) have been extensively used for insect pest management. Such proteins can be delivered by the bacterium itself (for use in organic agriculture or for suppression of mosquito larvae for example), and via transgenic plants. As Bt is a soil bacterium with pesticidal proteins detected on plant surfaces, these proteins have evolved to kill herbivorous insects that chew on plant material. There has been little selection for Bt proteins that kill sap-sucking or hemipteran insects that feed on plant sap. The hemipteran pests present a particular challenge as chemical insecticides represent the only tool available for management.
We are working on several approaches to improve the toxicity of Bt-derived pesticidal proteins against Hemiptera. Using the pea aphid as a model system, we examined the physiological bases for the lack of toxicity of selected Bt proteins. We demonstrated that different toxins behave differently in the aphid gut with some being unstable, and others binding but exerting only low levels of toxic action. Research on insect gut physiology to understand why toxins are or are not active when ingested led to a novel approach for retargeting of gut-active pesticidal proteins to species that are not normally susceptible, such as the pea aphid (Chougule et al., 2013).
We are currently adapting this strategy to target insects that lack susceptibility to Bt pesticidal proteins, or have developed resistance to them including the soybean aphid, Aphis glycines, and the Asian citrus psyllid, Diaphorina citri.
Related publications
Kuwar, S.S., Mishra, R., Banerjee, R., Milligan, J., Rydel, T., Du, Z., Xie, Z., Ivashuta, S., Kouadio, J.L., Meyer, J.M., Bonning, B.C. 2022 Engineering of Cry3Bb1 provides mechanistic insights toward countering western corn rootworm resistance. Curr. Res. Insect Sci. 2: 100033. https://doi.org/10.1016/j.cris.2022.100033
Banerjee, R., Flores-Escobar, B., Chougule, N.P., Canton, P.E., Dumitru, R., Bonning, B.C. 2022. Peptide mediated, enhanced toxicity of a bacterial pesticidal protein against southern green stink bug. Microbial Biotech 15: 2071-2082. doi: 10.1111/1751-7915.14030
Panneerselvam, S., Mishra, R., Berry, C., Crickmore, N., Bonning, B.C. 2022. BPPRC database: a web-based tool to access and analyze bacterial pesticidal proteins. Database (Oxford) baac022. doi: 10.1093/database/baac022
Mishra, R., Arora, A.K., Jiménez, J., Tavares, C.S., Banerjee, R., Panneerselvam, S., Bonning, B.C. 2022. Bacteria-derived pesticidal proteins active against hemipteran pests. Journal of Invertebrate Pathology 195: 107834 DOI: 10.1016/j.jip.2022.107834
A structure-based nomenclature for Bacillus thuringiensis and other bacteria-derived pesticidal proteins. Crickmore N, Berry C, Paneerselvam S, Mishra R, Connor TR, Bonning BC. 2020. Journal of Invertebrate Pathology 107438 (online in advance of publication). doi: 10.1016/j.jip.2020.107438
Streamlined phage display library protocols for identification of insect gut binding peptides highlight peptide specificity. Mishra R, Guo Y, Kumar P, Cantón PE, Tavares CS, Bannerjee R, Kuwar S, Bonning BC. 2021. Current Research in Insect Science 1, 100012. doi: 10.1016/j.cris.2021.100012
Toxicity of Bacillus thuringiensis-Derived Pesticidal Proteins Cry1Ab and Cry1Ba against Asian Citrus Psyllid, Diaphorina citri (Hemiptera). Fernandez-Luna MT, Kumar P, Hall DG, Mitchell AD, Blackburn MB, Bonning BC. Toxins (Basel). 2019 Mar 22;11(3). pii: E173. doi: 10.3390/toxins11030173.
Modification of Cry4Aa toward Improved Toxin Processing in the Gut of the Pea Aphid, Acyrthosiphon pisum. Rausch MA, Chougule NP, Deist BR, Bonning BC. PLoS One. 2016 May 12;11(5):e0155466. doi: 10.1371/journal.pone.0155466. eCollection 2016.
Bt toxin modification for enhanced efficacy. Deist BR, Rausch MA, Fernandez-Luna MT, Adang MJ, Bonning BC. Toxins (Basel). 2014 Oct 22;6(10):3005-27. doi: 10.3390/toxins6103005. Review.
Chougule, N.P., Li, H. Liu, S., Narva, K.E., Meade, T., Bonning, B.C. 2013. Retargeting of Bt toxins against hemipteran insect pests. Proc. Natl. Acad. Sci. USA
Chougule, N.P. and Bonning, B.C. 2012. Toxins for transgenic resistance to hemipteran pests. Special issue of the online journal Toxins “Insecticidal Toxins” 4(6), 405-429 doi:10.3390/toxins4060405
Li, H., Chougule, N.P., Bonning, B.C. 2011. Interaction of the Bacillus thuringiensis delta endotoxins Cry1Ac and Cry3Aa with the gut of the pea aphid, Acyrthosiphon pisum (Harris). J. Invertebr Pathology 107: 69-78.
Insect Gut Physiology
Similar to the lack of efficacy of Bt pesticidal proteins against aphids, improved understanding of the enzymatic environment encountered by protein- or RNA-based control technologies can provide leads for improvement of- or delivery systems for- pest management agents. In this regard, stink bugs present a particular challenge.
Stink bugs are hemipteran pests that deleteriously impact 12 major agricultural crops across the globe. Stink bugs rely on a battery of digestive enzymes for extra-oral as well as gut-based digestion of plant tissues. Stink bug salivary enzymes are released into the plant and the partially digested proteins ingested for further degradation in the gut. With funding from CAMTech, we characterized both the proteases and nucleases in the gut and salivary gland of the southern green stink bug, Nezara viridula (Linnaeus) and the brown marmorated stink bug, Halyomorpha halys and enzyme coding sequences were identified. Biochemical characterization showed that the proteases in the gut and saliva differ radically in composition allowing for a two-pronged approach for digestion of plant material. Nuclease activity is abundant in the saliva of both stink bug species. These enzymes present a major challenge for the stability of putative stink bug control agents.
Related publications
Jiménez, J., Mishra, R., Wang, X., Magee, C.M., Bonning, B.C. 2024. Composition and abundance of midgut surface proteins in two major hemipteran vectors of plant viruses, Bemisia tabaci and Myzus persicae. Arch. Insect Biochem. Physiol. 116(3):e22133. doi: 10.1002/arch.22133
Tavares, C.S., Mishra, R., Ghobrial, P.N., Bonning, B.C. 2022. Composition and abundance of midgut surface proteins in the Asian citrus psyllid, Diaphorina citri. J. Proteomics 261:104580 doi: 10.1016/j.jprot.2022.104580
Extra oral digestion: outsourcing the role of the hemipteran midgut. Canton PE, Bonning BC. 2020. Current Opinion in Insect Science 41: 86-91. doi: 10.1016/j.cois.2020.07.006
Transcription and activity of digestive enzymes of Nezara viridula maintained on different plant diets. Canton PE, Bonning BC. 2020. Frontiers in Physiology 10, 1553. doi: 10.3389/fphys.2013.01553
Transcription and activity of digestive enzymes of Nezara viridula maintained on different plant diets. 2019. PE Cantón, BC Bonning. doi: 10.3389/fphys.2019.01553
The principal salivary gland is the primary source of digestive enzymes in the saliva of the brown marmorated stink bug, Halyomorpha halys. Liu S, Bonning BC. 2019. Frontiers in Physiology 10, 1553. doi: 10.3389/fphys.2019.01553
Proteases and nucleases across midgut tissues of Nezara viridula (Hemiptera: Pentatomidae) display distinct activity profiles that are conserved through life stages. 2019. PE Cantón, BC Bonning. Journal of insect physiology 119, 103965. doi: 10.1016/j.jinsphys.2019.103965.
Tissue-specific transcription of proteases and nucleases across the accessory salivary gland, principal salivary gland and gut of Nezara viridula. Liu S, Lomate PR, Bonning BC. Insect Biochem Mol Biol. 2018 Dec;103:36-45. doi: 10.1016/j.ibmb.2018.10.003. Epub 2018 Oct 21.
Proteases and nucleases involved in the biphasic digestion process of the brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). Lomate PR, Bonning BC. Arch Insect Biochem Physiol. 2018 Jul;98(3):e21459. doi: 10.1002/arch.21459
Distinct properties of proteases and nucleases in the gut, salivary gland and saliva of southern green stink bug, Nezara viridula. Lomate PR, Bonning BC. Sci Rep. 2016 Jun 10;6:27587. doi: 10.1038/srep27587.