common name: yellow Brazilian peppertree leaf-galler (suggested common name)
scientific name: Calophya latiforceps Burckhardt (Insecta: Hemiptera: Calophyidae: Calophyinae)
Introduction - Distribution - Description - Life Cycle and Biology - Host Plants - Economic Importance - Selected References
Introduction (Back to Top)
In the late 1970s, Brazilian peppertree, Schinus terebinthifolia Raddi (Sapindales: Anacardiaceae), was targeted for classical biological control in Florida because its invasive properties (see Host Plants) are consistent with escape from natural enemies (Williams 1954), and there are no native Schinus spp. in North America. The lack of native close relatives should minimize the risk of damage to non-target plants from introduced biological control agents (Pemberton 2000).
Several exploratory surveys for natural enemies of Brazilian peppertree have been conducted in South America (Hight et al. 2002, Cuda et al. 2006, McKay et al. 2009, Wheeler et al. 2016). In 2010, Calophya latiforceps Burckhardt, a new species of leaflet galling psyllid we are calling the yellow Brazilian peppertree leaf-galler (Figure 1), was discovered attacking Brazilian peppertree in the northern state of Bahia, Brazil (Burckhardt et al. 2011).
Figure 1. Leaves of Brazilian peppertree, Schinus terebinthifolia, attacked by developing nymphs Calophya latiforceps Burckhardt. Photograph by Rodrigo Diaz, LSU AgCenter, Baton Rouge, LA.
The genus Calophya Löw is a predominantly New World, Oriental and East Palaearctic genus of 59 described species that are mostly associated with Anacardiaceae and other Sapindales. Among the psyllids developing on Schinus, there are 14 described Calophya species, which all induce galls (Burckhardt and Basset 2000). These authors showed that several Calophya spp. are monophagous, meaning they are restricted to a single host plant species.
Distribution (Back to Top)
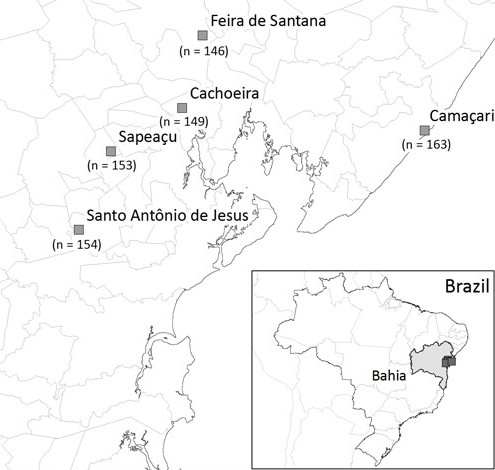
Calophya latiforceps has been collected from only a few localities in northeastern Brazil in the Bahia state. (Figure 2) (Burckhardt et al. 2011).
Figure 2. Distribution of Calophya latiforceps Burckhardt. Map produced by Abhishek Mukherjee, Indian Statistical Institute, Kolkata, India.
Description (Back to Top)
Eggs: Eggs are laid along the margins and veins of new leaf flushes. Newly laid eggs are oblong in shape, white in color (< 3 days old) and turn black before nymphal hatching (Figure 3). Egg length is probably similar to that of Calophya terebinthifolii Burckhardt & Bassett, which is 0.212 ± 0.002 mm (Christ et al. 2013).
Figure 3. Eggs of Calophya latiforceps Burckhardt. Photograph by Rodrigo Diaz, LSU AgCenter, Baton Rouge, LA.
Nymphs: The first instar nymphs are often referred to as crawlers. Subsequent instars are not mobile, they are bright yellow and secrete waxy droplets (Figures 4). When removed from the leaf, the fifth instar resembles a half-sphere. Length and width of the 5th instar are 0.82 mm and 0.79 mm, respectively.
Figure 4. Late instar nymph of Calophya latiforceps Burckhardt. Photograph by Rodrigo Diaz, LSU AgCenter, Baton Rouge, LA.
Adults: Adults are bright yellow and are found on new flushes of Brazilian peppertree. Females are larger in size than males; 1.59 ± 0.01 mm in length compared to 1.48 ± 0.01 mm. The eyes are greyish in color. The antennae are dirty yellowish basally and gradually become darker towards the apex. The tibiae and tarsi are greyish brown. The forewing has yellow or light brown veins, and the membrane is colorless and transparent. The sex of the adults can be distinguished by the shape of the tip of the abdomen, which is pointed in females (Figures 5 and 6).
Figure 5. Male of Calophya latiforceps Burckhardt. Photograph by Rodrigo Diaz, LSU AgCenter, Baton Rouge, LA.
Figure 6. Female of Calophya latiforceps Burckhardt. Photograph by Rodrigo Diaz, LSU AgCenter, Baton Rouge, LA.
Life Cycle and Biology (Back to Top)
According to Diaz et al. (2014a), newly emerged adults are pale green in color and remain inactive for ~30 minutes on the leaflet from which they emerged. Adults remain on the plant during the cooler hours of the morning or late evening. Adults are poor fliers and move < 30 cm in random jumps. Long distance dispersal is presumably aided by wind currents. Groups of adults feed and search for mates on new leaf flushes, and copulation occurs a few hours after emergence; mating lasts 3 to 5 min. Adults live on average 9.3 ± 0.6 d (range 6.3 to 12 days).
Females oviposit on new leaflets (<2 cm long); eggs are laid individually along the leaflet margins and veins as well as along leaf petioles. After eclosion, first instars (crawlers) walk slowly on the upper side of leaflets and settle after a couple of hours. Most of the crawlers settle on the same leaflet where the eggs were laid.
Twenty-four hours after settling, a yellow halo in the plant tissue appears around the nymphs. Susceptible plants respond to nymphal feeding by forming a slight depression whereas resistant plants respond by killing the plant cells at the point of feeding (Figure 1). This hypersensitive response was noticeable two or three days after nymphs settled and resulted in 100% mortality of the psyllids (Diaz et al. 2014a). Surviving nymphs increase in size rapidly as they mature. There are five instars and total developmental time to the adult stage ranges from 35 to 53 days (average 38.6 days).
Host Plants (Back to Top)
The only known host plant for Calophya latiforceps Burckhardt is Brazilian peppertree, Schinus terebinthifolia. Quarantine host range studies on 99 plant taxa (species, varieties and hybrids) from 43 families showed that the psyllid was only able to colonize Brazilian peppertree (Diaz et al. 2014b). On average, 77% of Florida Brazilian peppertrees are susceptible to attack by Calophya latiforceps Burckhardt.
Brazilian peppertree is a perennial woody plant native to Brazil, Argentina, and Paraguay that has become one of the most invasive upland weeds in Florida (Cuda et al. 2006). Originally introduced to Florida in the 1840s as an ornamental (Mack 1991), it escaped cultivation in the 1950s (Morton 1978, Austin and Smith 1998), and invaded disturbed sites, natural communities, and environmentally sensitive areas such as the Everglades National Park (Ewel et al.1982). In central and south Florida, Brazilian peppertree occupies more than 2,833 km2 (Cuda et al. 2006, Manrique et al. 2013), and outcompetes native species by exhibiting fast growth, prolific seed production, and vigorous resprouting.
Brazilian peppertree tolerates a wide range of growing conditions including high salinity, moisture, and shade (Ewel 1979; Ewe and Sternberg 2005, 2007). In FY 2015 – 2016, the South Florida Water Management District spent over $2.6 million controlling Brazilian peppertree on district lands (Rodgers et al. 2017). Because of its severe environmental impacts, Brazilian peppertree is listed as a prohibited plant in Florida (UF/IFAS Assessment).
Economic Importance (Back to Top)
Calophya latiforceps Burckhardt is highly host specific, and is capable of decreasing photosynthesis, inhibiting growth and inducing leaf abscission (Prade et al. 2016). Nymphal feeding habits are detrimental to the plant whereas the damage by the adults is insignificant. Injury to the plant results from the injection of toxic saliva, which causes toxemia in the host. Psyllid damage may be localized and/or systemic and may manifest as necrosis, leaf rolling and changes in leaf color, withering, and gall formation (Hodkinson 1974). Calophya latiforceps Burckhardt was recommended for release in Florida for biological control of Brazilian peppertree in April 2016. If the insect establishes in Florida post-release, it will contribute to the sustainable management of Brazilian peppertree, while posing minimal risk to non-target species.
Acknowledgements
We appreciate the contributions and editorial comments by Dr. Rodrigo Diaz, Department of Entomology, Louisiana State University, Baton Rouge, LA, and Dr. Daniel Burckhardt, Naturhistorisches Museum, Augustinergasse 2, Basel, CH-4001, Switzerland.
Selected References (Back to Top)
- Austin DF, Smith E. 1998. Pine Rockland Plant Guide: A Field Guide to the Plants of South Florida’s Pine Rockland Community. Dade County Environmental Resources Management, Miami, Florida.
- Burckhardt D, Bassett Y. 2000. The jumping plant-lice (Hemiptera, Psylloidea) associated with Schinus (Anacardiaceae): Systematics, biogeography and host plant relationships. Journal of Natural History 34: 57-155.
- Burckhardt D, Cuda JP, Manrique V, Diaz R, Overholt WA, Williams DA, Christ LR, Vitorino MD. 2011. Calophya latiforceps, a new species of jumping plant lice (Hemiptera: Calophyidae) associated with Schinus terebinthifolius (Anacardiaceae) in Brazil. Florida Entomologist 94: 489-499.
- Christ LR, Cuda JP, Overholt WA, Vitorino MD, Mukherjee A. 2013. Biology, host preferences, and potential distribution of Calophya terebinthifolii (Hemiptera: Calophyidae), a candidate for biological control of Brazilian peppertree, Schinus terebinthifolius, in Florida. Florida Entomologist 96: 137-147.
- Cuda, JP, Ferriter AP, Manrique V, Medal JC. (eds.). 2006. Florida’s Brazilian peppertree management plan: Recommendations from the Brazilian Peppertree Task Force. 2nd ed., Florida Exotic Pest Plant Council. Available via Center for Invasive Species and Ecosystem Health at the University of Georgia.
- Diaz R, Moscoso D, Manrique V, Williams D, Overholt WA. 2014a. Native range density, host utilization and life history of Calophya latiforceps (Hemiptera: Calophyidae): An herbivore of Brazilian peppertree (Schinus terebinthifolia). Biocontrol Science & Technology 24: 536-553.
- Diaz R, Manrique V, Munyaneza JE, Sengoda VG, Adkins S, Hendricks K, Roberts PD, Overholt WA. 2014b. Host specificity testing and examination for plant pathogens reveal that the gall-inducing psyllid Calophya latiforceps is safe to release for biological control of Brazilian peppertree. Entomologia Experimentalis et Applicata 1-14 DOI: 10.1111/eea.12249.
- Ewe SML, Sternberg LSL. 2005. Growth and gas exchange patterns of the invasive exotic Schinus terebinthifolius contrasted with native species under controlled saline conditions. Trees 19(2): 119-128.
- Ewe SML, Sternberg LSL. 2007. Water uptake patterns of an invasive exotic plant in coastal saline habitats. Journal of Coastal Research 23: 255-264.
- Ewel JJ. 1979. Ecology of Schinus, pp. 7-22 in Workman R (ed.), Schinus- Technical Proceedings of Techniques for Control of Schinus in South Florida: A Workshop for Natural Area Managers, 2 December 1978. The Sanibel Captiva Conservation Foundation, Inc., Sanibel, Florida.
- Ewel JJ, Ojima DK, Debusk W.1982. Schinus in successional ecosystems of Everglades National Park. South Florida Res. Cent. Rep. T-676. Everglades National Park, National Park Service, Homestead, Florida.
- Hight SD, Cuda JP, Medal JC. 2002. Chapter 24, Brazilian Peppertree, pp. 311-321, in Van Driesche R, Blossey B, Hoddle M, Lyon S, Reardon R (eds.), Biological Control of Invasive Plants in the Eastern United States. USDA Forest Service Publication FHTET-2002-04, Morgantown, WV.
- Hodkinson ID. The biology of the Psylloidea (Homoptera): A review. Bulletin of Entomological Research 64: 325-339.
- Mack RN. 1991. The commercial seed trade: An early disperser of weeds in the United States. Economic Botany 45: 257-273.
- Manrique V, Cuda JP, Overholt WA. 2013. Brazilian peppertree: A poster child for invasive plants in Florida landscapes. Journal of Florida Studies 1(2): 1-14.
- McKay F, Oleiro M, Walsh GC, Gandolfo D, Cuda JP, Wheeler GS. 2009. Natural enemies of Brazilian peppertree (Schinus terebinthifolius: Anacardiaceae) from Argentina: Their possible use for biological control in the USA. Florida Entomologist 92: 292-303.
- Morton JF. 1978. Brazilian pepper: Its impact on people, animals and the environment. Economic Botany 32: 353-359.
- Pemberton RW. 2000. Predictable risk to native plants in weed biological control. Oecologia 125: 489-494.
- Prade P, Diaz R, Vitorino MD, Cuda JP, Kumar P, Gruber B, Overholt WA. 2016. Galls induced by Calophya latiforceps Burckhardt (Hemiptera: Calophyidae) reduce leaf performance and growth of Brazilian peppertree. Biocontrol Science and Technology 26: 23-34.
- Rodgers L, Mason C, Bodle M, Brown R, Ellen A, Tipping P, Rochford M, Mazzotti F, Peters A, Renda M, Beeler C, Ketterlin-Eckles J, Laroche F, Segura C, Serbesoff-King K. 2017. Chapter 7: Status of nonindigenous species, pp. 1-58, in 2017 South Florida Environmental Report, Vol. I. The South Florida Environment. South Florida Water Mgt. District, West Palm Beach, FL.
- Wheeler GS, Mc Kay F, Vitorino MD, Manrique V, Diaz R, Overholt WA. 2016. Biological control of the invasive weed Schinus terebinthifolia (Brazilian peppertree): A review of the project with an update on the proposed agents. Southeastern Naturalist 15(sp8): 15-34.
- Williams JR. 1954. The biological control of weeds, pp. 95-98. In: Rept Sixth Commonwealth Entomology Congress, London, UK.