common name: squash vine borer
scientific name: Melittia cucurbitae (Harris) (Insecta: Lepidoptera: Sesiidae)
Introduction - Synonymy - Distribution - Description - Life Cycle - Host Plants - Damage - Management - Selected References
Introduction (Back to Top)
Squash vine borer, Melittia cucurbitae (Harris), is a diurnal (active during the day) moth species (Figure 1). The larvae complete their growth and development on wild and domesticated species of the genus Cucurbita. This insect was once considered a nuisance to commercial growers and a problem to home growers of cucurbits. However, with the expansion of cucurbit production in the United States (U.S.) over the last decade, the squash vine borer has become a pest of economic importance (Brust 2010).
Figure 1. Adult squash vine borer, Melittia cucurbitae (Harris). Photographs by Lyle J. Buss, University of Florida.
Synonymy (Back to Top)
Aegeria cucurbitae Harris, 1828
Melittia satyriniformis (Hübner, [1827-1831])
Trochilium ceto (Westwood, 1848)
Melittia amoena (Edwards, 1882)
(From Beutenmuller W. 1897, Pühringer F, Kallies A. 2004, and ITIS 2014)
Distribution (Back to Top)
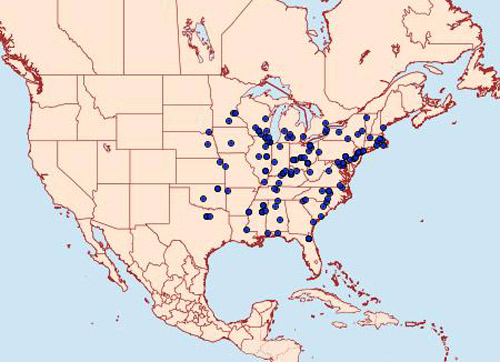
Squash vine borer is native to North America and is distributed throughout the eastern U.S. (Figure 2), southeastern Canada, and from Mexico to near Guatemala (Klun et al. 1990, Jackson et al. 2005). In Florida, the insect is common in most home gardens.
Figure 2. Reported distribution of the squash vine borer, Melittia cucurbitae (Harris), in the United States. Map by moths photographers group, Mississippi State University.
Description (Back to Top)
Eggs: The eggs of the squash vine borer are laid singly on the lower part of the main stem of the host plant, as well as on the leaf stalks, leaves, and fruit buds. Some eggs are laid in the cracks in the soil near the base of the plant (Canhilal et al. 2006). Eggs are dark to reddish brown, ovoid but slightly flattened, and small; they measure approximately 1 mm in length and 0.85 mm in width (Figure 3, Capinera 2008, Bauernfeind and Nechols 2005).
Figure 3. In the center of the red circle is one egg of the squash vine borer, Melittia cucurbitae (Harris). Photograph by Theresa Friday, University of Florida.
Larvae: The larvae are white, with a darkened head capsule, three pairs of thoracic legs, and five pairs of prolegs (peg-like legs on the abdomen of a larva). Newly emerged larvae are 1.5 to 2 mm long, tapered on the posterior end, and covered with numerous large hairs. As the larva matures, it develops a dark thoracic shield, loses its tapered shape and hairy appearance, and grows to a length of approximately 25 mm (Figure 4, Bauernfeind and Nechols 2005, Capinera 2008). Late instar larvae burrow into the soil before pupation.
Figure 4. Larva of the squash vine borer, Melittia cucurbitae (Harris). Photograph by James Castner, University of Florida.
Pupae: The pupae of the squash vine borer are mahogany brown, approximately 14 mm long, and enclosed in silk cocoons (Bauernfeind and Nechols 2005).
Adults: The adult squash vine borer is thought to resemble a wasp (Figure 1). They are approximately 16 mm in length, with a wingspan range of 25 to 38 mm. The front wings are covered with scales that give them a metallic green to black sheen. Large portions of the hind wings lack scales, making them look clear. The abdomen is covered with conspicuous orange to reddish hairs, punctuated dorsally with black dots (Figure 1). The hind legs are covered with long black hairs inside and orange hairs outside (Bauernfeind and Nechols 2005). Females are larger and less colorful than males, with wider abdomens.
Unlike most moth species, which are active at night and have scaly wings, squash vine borer adults (and many members of the family Sesiidae) are active during the day, rest in the evenings, and have clear wings. The adults can be observed during the day feeding on nectar (Canhilal et al. 2006).
Figure 5. Adult squash vine borer, Melittia cucurbitae (Harris). (1) Abdomen covered by orange to reddish hairs and punctuated with black dots dorsally, (2) Front wings covered by scales that give them a metallic green to black sheen, (3) Hind legs covered with long black hairs inside and orange hairs outside. Photograph by Lyle J. Buss, University of Florida.
Life Cycle (Back to Top)
Females usually lay eggs about one day after emergence, and each can produce 150 to 200 eggs in her lifetime (Capinera 2008). The eggs hatch in eight to 14 days (Canhilal et al. 2006). Newly emerged larvae burrow into vines, where they feed on the host plant tissue. The larvae develop through four instars in four to six weeks. The late instar larvae drop to the ground and burrow 25 to 50 mm into the soil to pupate. The pupae complete development in 14 to 30 days. In the field, the average lifespan of the females is about five days and that of males is about three days (Canhilal et al. 2006).
The squash vine borer has two overlapping generations per year in the southern U. S. but only one generation in the northern U.S. (Jackson et al. 2005, Canhilal et al. 2006). In intermediate states like Ohio, some of the borer population undergoes diapause after one generation, but most complete two generations a year (Capinera 2008). In Florida and other southern states, squash vine borer overwinters at the end of October as mature larvae or pupae enclosed in cocoons 25 to 50 mm below the soil surface. Adults emerge from cocoons from mid-May to late October (Canhilal et al. 2006).
Host Plants (Back to Top)
The host range of the squash vine borer is restricted to the genus Cucurbita. This insect attacks both cultivated and wild cucurbits, but prefers host plant species with large hollow stems to those with narrow stems (Howes and Rhodes 1973). It is a major pest of summer squash (Cucurbita pepo L.) and pumpkin (Cucurbita maxima Duchesne), but a minor pest of cucumber (Cucumis sativus L.), watermelon (Citrullus lanatus (Thunb.)), cantaloupe (Cucumis melo L.), and Mexican gourd (Curcubita ficifolia Bouché) (Howe 1950). Squash and pumpkin varieties differ widely in their susceptibility to damage by squash vine borers (Gould 2013).
Economic Importance (Back to Top)
Attack by squash vine borer on pumpkin and squash crops has been reported to cause over 25% yield loss in commercial farms (Ghidiu 2005, Bauernfeind and Nechols 2005). In home gardens, the loss can be as high as 100% because the pest population is usually concentrated on a limited number of plants (Pearson 1995). This makes squash vine borer an insect pest of great economic importance in the United States. In 2012, annual production of squash and pumpkins in the U.S. was valued at approximately $400 million. The value of pumpkins produced was $149 million, and that of squash was $249 million (National Agricultural Statistics Service 2013).
Damage (Back to Top)
Squash vine borer larvae tunnel into vines to feed. The larval feeding destroys plant vascular tissue and interrupts the flow of water and nutrients from the roots to the developing fruit. This interruption of water and nutrient flow causes sudden localized wilting of the affected plants, usually observed during the middle of the day when temperature rise, while the surrounding uninfested plants look healthy.
Inspection of infested plants can reveal an entry hole where the larva entered the plant surrounded by green frass (sawdust-like excrement) (Brust 2010) (Figure 5). Dissecting the infested plants will reveal the squash borer larvae.
A single larva feeding on a pumpkin plant can reduce yield by up to 4%, three to four larva per plant are capable of causing a yield loss of more than 20%. Six or more larvae per plant are enough to severely damage or kill a plant (Brust 2010). In addition, the larval feeding predisposes the host plant to infection by opportunistic plant pathogens.
Figure 6. Squash vine borer damage, Melittia cucurbitae (Harris), on squash. Photograph by James Castner, University of Florida.
Management (Back to Top)
Cultural control: To reduce the population density of the squash vine borer the following season, field sanitation is recommended in fields with known infestation. Vines should be collected and discarded, or plowed under as soon as the crop is harvested. Plowing after harvest is particularly recommended, as it will destroy the overwintering stage of the insect in the soil.
Fields with a prior infestation of squash vine borer should be planted with least preferred varieties such as Waltham butternut. Planting of least preferred varieties helps growers reduce the frequency of pesticide application (Gould 1958). Alternatively, planting dates can be staggered to allow some crops to escape periods of heavy oviposition.
To reduce the pest density on a cucurbit crop, lightweight row covers can be used so that the adult squash vine borer can lay eggs on them and not on the main crop. During the flowering period, the row covers should be removed to allow pollinators to reach the main crop. Alternatively, a high-preferred variety trap crop can be planted around the main crop to divert insect pressure from the main crop. This method only works if the main crop is a less-preferred variety (Boucher and Durgy 2002).
As a cucurbit crop is growing, any plants killed by squash vine borer should be uprooted and destroyed to prevent the insect from completing its life cycle. However, this method is labor intensive and may only be applicable to home gardening. Moist soil should be heaped over stem joints to promote root development, so that in the event squash vine borer damages the main stem base new roots will continue to nourish the plant.
Chemical control: Chemical control is the main method of controlling squash vine borer in the United States. Several insecticides are effective against the squash vine borer but only with timely application (Capinera 2008). Entomopathogenic nematodes (families Steinernematidae and Heterorhabditidae) have been used to manage squash vine borer. The nematodes provided the same level of control as a conventional pesticide application (Canhilal and Carner 2006). Insecticide (chemical or biological) application should be done early in the season to kill larvae as they hatch from the eggs, before they burrow into the vines. Delayed application is ineffective because once the larvae have tunneled into the vines they cannot be reached by most insecticides (Ghidiu 2005).
Growers can employ either of two main chemical control strategies: The first strategy is based on weekly application of insecticide regardless of whether the squash vine borer has been detected or not. Spraying starts immediately after the vines begin to run and is continued for four to six weeks (Brust 2010). This strategy is relatively simple to implement but the weekly application of insecticides results in excessive chemical use that can harm non-target organisms. In the second strategy, the crop is sprayed only after the population of squash vine borer has reached the economic threshold (Brust 2010).
The economic threshold for squash vine borer is two or more individuals found per 20 m of crop row or caught in pheromone traps per week (Brust 2010). The squash vine borer population is monitored early in the season by scouting and pheromone trapping of adult moths. This second strategy is as effective as the first strategy described earlier, but it requires less pesticide. However, it is relatively labor intensive, hard to implement, and is only economically feasible when dealing with more than 5 acres of crop (Brust 2010). To protect pollinators from pesticides, any pesticide application during the flowering period should be done late in the day when pollinator activity is at a minimum (Capinera 2008).
Selected References (Back to Top)
- Bauernfeind RJ, Nechols JR. 2005. Squash bugs and squash vine borers. Kansas State University Agricultural Experiment Station and Cooperative Extension Service Bulletin MF-2508. (31 March 2020).
- Beutenmuller W. 1897. Note on Melittia satyriniformis Hübner. Journal of the New York Entomological Society 5: 34-35.
- Boucher TJ, Durgy R. 2002. Effect of spinosad on winter squash pests, 2001. Arthropod Management Tests 27: E84.
- Brust GE. 2010. Squash vine borer (Lepidoptera: Sesiidae) management in pumpkin in the mid-Atlantic. Journal of Applied Entomology 134: 781-788.
- Canhilal R, Carner GR. 2006. Efficacy of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) against the squash vine borer, Melittia cucurbitae (Lepidoptera: Sesiidae) in South Carolina. Journal of Agricultural and Urban Entomology 23: 27-39.
- Canhilal R, Carner GR, Griffin RP, Jackson DM, Alverson DR. 2006. Life history of the squash vine borer, Melittia cucurbitae (Harris) (Lepidoptera: Sesiidae) in South Carolina. Journal of Agricultural and Urban Entomology 23: 1-6.
- Capinera JL. 2008. Squash vine borer, Melittia cucurbitae (Harris) (Lepidoptera: Sesiidae). In Encyclopedia of Entomology (pp. 3533-3536). Springer Netherlands.
- Ghidiu G. 2005. Squash vine borers. New Brunswick (NJ): Rutgers NJAES Cooperative Extension. njaes.rutgers.edu/pubs/download free.asp?strPubID=FS229 (31 March 2020).
- Gould GE. 1958. Varietal susceptibility of cucurbits to cucumber beetle attack. Proceedings of the Indiana Academy of Science 68: 186-189.
- Howe WL. 1950. Biology and host relationship of squash vine borer. Journal of Economic Entomology 43: 480-483.
- Howe WL, Rhodes AM. 1973. Host relationships of the squash vine borer, Melittia cucurbitae with species of Cucurbita. Annals of the Entomological Society of America 66: 266-269.
- Jackson DM, Canhilal R, Carner GR. 2005. Trap monitoring squash vine borers in Cucurbits 1, 2. Journal of Agricultural and Urban Entomology 22: 27-39.
- Klun JA, Schwarz M, Leonardt BA, Cantelo WW. 1990. Sex pheromone of the female squash vine borer. Journal of Entomological Science 25: 64-72.
- National Agricultural Statistics Service. 2013. Vegetables 2012 summary. United States Department of Agriculture. (31 March 2020).
- Pearson GA. 1995. Sesiid pheromone increases squash vine borer (Lepidoptera: Sesiidae) infestation. Environmental Entomology 24: 1627-1632.
- Pühringer F, Kallies A. 2004. Provisional checklist of the Sesiidae of the world (Lepidoptera: Ditrysia). Mitteilungen der Entomologischen Arbeitsgemeinschaft Salzkammergut 4: 1-85.