common name: Florida woods cockroach (aka palmetto bug)
scientific name: Eurycotis floridana (Walker) (Insecta: Blattodea: Blattidae: Polyzosteriinae)
Introduction - Synonymy - Distribution - Description - Biology and Life Cycle - Feeding and Habitat - Defenses - Management - Selected References
Introduction (Back to Top)
The Florida woods cockroach, Eurycotis floridana (Walker), is native to the southeastern United States, where it is often referred to as a palmetto bug. It is a peridomestic organism that can be found in structures near the home, such as storage areas, greenhouses, or shelter boxes for other structures such as water pumps. It is also an occasional invader of the home, but lacks effective flight wings and is slow moving. Its presence in the home is typically a result of being transported indoors on another item, such as firewood (Smith & Whitman 1992). Eurycotis floridana has not been shown to commonly colonize inside structures, with limited instances of attic breeding (Atkinson et al. 1990). This species also shows no preferences for consuming waste or debris of urban consequence (Cornwell 1968). As a result, the species tends to remain sylvan with overlap into peridomestic areas for protection from sunlight or foraging for food (Atkinson et al. 1990, Cornwell 1968, Young & Cantrall 1958).
Figure 1. Lateral-dorsal view of an adult Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
The genus Eurycotis has one other member present in the United States that is confined to the Florida Keys, Eurycotis lixa Rehn (Gurney 1959). With respect to this relative, the Florida woods cockroach is the northernmost representative of its neotropical genus (Atkinson et al. 1990).
Synonymy (Back to Top)
Eurycotis floridana was originally described as Periplaneta floridana (Walker 1868), as well as the following synonyms:
- Periplaneta semipicta (Walker 1868)
- Platyzosteria ingens (Scudder 1877)
- Platyzosteria sabaliana (Scudder 1877)
(Atkinson et al. 1990, Rehn 1902).
Eurycotis floridana has no official common name as is sanctioned by the Entomological Society of America, but is known by the common names Florida woods cockroach, Florida stinkroach, skunk cockroach, and palmettobug. However, palmettobug may also describe any large cockroach that may frequently be encountered as a peridomestic pest, and has included several members of Eurycotis and Periplaneta (Gurney & Walker 1976) such as the American cockroach, Periplaneta americana (Linnaeus), and the smokybrown cockroach, Periplaneta fuliginosa (Serville).
Distribution (Back to Top)
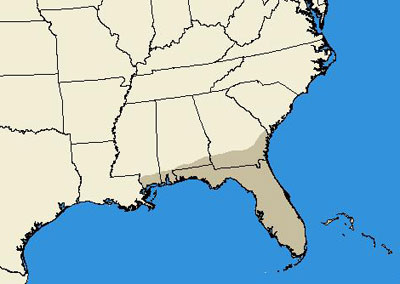
Eurycotis floridana occurs in a limited southeastern range in the United States. Records indicate presence in coastal Georgia, Alabama, and Mississippi, and throughout most of Florida, with specific records in the counties of Alachua, Broward, Charlotte, Citrus, Clay, Collier, Dade, Duval, Escambia, Levy, Highlands, Hillsborough, Polk, Putnam, Miami-Dade and Monroe (Hagenbuch et al. 1988, Hebard 1917, Atkinson et al. 1990, Dakin et al. 1970, Smith & Whitman 1992).
Figure 2. State level distribution map of Eurycotis floridana (Walker).(From Hagenbuch et al. 1988, Hebard 1917, Atkinson et al. 1990, Dakin et al. 1970 and Smith & Whitman 1992).
Description (Back to Top)
Adults: The adult is distinctively large in size, ranging from 30—40 mm (1.20—1.60 in) long and up to 24 mm (1.00 in) wide. The body ranges in color from reddish brown to nearly black. This species also lacks developed wings in either sex, instead having reduced wing pads and absent hind wings. The wings form short leathery pads, referred to as tegmina, which barely meet on the midline of the body and do not extend to cover any portion of the abdomen.
Figure 3. Vestigial wing formation on an adult Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
The adults have well developed cerci and tooth-like projections at the edges of the dorsal segments that approach the cerci.
Figure 4. Dorsal view of cerci and posterior sclerites of an adult Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
Males and females are not considered dimorphic. However, the size of the cockroach makes it easier to see differences in the posterior structure of the body segments, most notably the rectal area. Females have a terminal body segment that is fused together and appears wider than the previous segments, with a v-shaped notch. Males lack this larger segment, instead having filaments symmetrically placed within the interior zone of cerci, but before the rectal opening.
Figure 5. Close up of terminal sclerites located posteriorly; note the "V" notch on an adult female Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
Figure 6. Close up of terminal sclerites located posteriorly, note the symmetrical filaments, on an adult male Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
Adult lifespan exceeds one year. Compared to other roaches, the Florida woods cockroach is relatively slow moving and not quick to run away from disturbance. This could be due to its large size or lack of urgency due to the presence of defensive chemicals.
Figure 7. Ootheca as it is being formed by an adult female Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
Eggs: Eggs are deposited into an ootheca which forms while attached to the adult female. The ootheca will initially appear pale as it lacks a hardened cuticle. Over time, the casing will darken from reddish brown to black as the cuticle hardens. Size ranges between 13—16 mm (0.51 —0.63 in) and the dorsal edge of the ootheca will have 18—24 sequential saw-like teeth. The ootheca may be dropped prematurely if the adult is startled, harassed or in danger.
Figure 8. Fully developed and detached ootheca of the Florida woods cockroach, Eurycotis floridana (Walker). Photograph by Christopher S. Bibbs, University of Florida.
Nymphs: Eurycotis floridana nymphs are reddish brown to black in color. Nymphs average seven molts during pre-adult growth. Late nymphal stages commonly have yellow thoracic margins.
Figure 9. Nymph of the Florida woods cockroach, Eurycotis floridana (Walker), showing yellow margins on the pro-, meso-, and metanota. Photograph by Christopher S. Bibbs, University of Florida.
Figure 10. Nymph of the Florida woods cockroach, Eurycotis floridana (Walker), without yellow margins on the pro-, meso-, and metanota. Photograph by Christopher S. Bibbs, University of Florida.
Biology and Life Cycle (Back to Top)
Eggs within the ootheca take approximately 48 days to hatch at 30—36°C. Once hatched, the nymphs undergo six to eight molts before reaching adulthood within 100—120 days. This time period can be accelerated if nymphs are reared in groups (Willis et al. 1958). Egg to adult maturation time is estimated to be 150 days (Smith & Whitman 1992).
Regardless of copulation, the adult female produces her first ootheca after 55 days in adulthood. If not fertilized, the species has limited parthenogenic development, with 60% or less of the eggs being viable and few individuals successfully hatching (Cornwell 1968). Individuals that hatch parthenogenically will not mature to adulthood (Robinson 2005). The presence of males is still necessary because there is no recruitment without fertilization. To facilitate mating, males will use volatile phermonal signals to attract a female over relatively large areas (Barth 1968, Farine et al. 1994). Once the sexual encounter is initiated, males will begin a courtship dance that involves mutual investigation and small amplitude side to side motions that is also characteristic of Periplaneta aggression responses (Barth 1970).
Following courtship and acceptance, the male exposes glands on the dorsal surface of his abdomen by extending the body segments and revealing gaps between the second and third plates and the seventh and eighth plates. These glands are lined with setae that present the secretions as an offering to the female to lure her into an appropriate copulatory position (Barth 1968, Farine et al. 1994). After mounting and successful attachment, the individuals will assume an opposed posture with the abdominal tips connecting and the heads facing away from each other (Barth 1968). This courtship and copulatory procedure in E. floridana is unique in the courtship movements and length of interaction when compared to other blattids (Barth 1968).
The adult female often glues her ootheca onto the substrate in a potential dwelling, such as a decaying log (Robinson 2005). The ootheca has roles in gas exchange, mechanical protection and osmoregulation using the substrate; substrates with excessive water retention will lyse the ootheca from extended water absorption and substrates with insufficient water retention will cause the ootheca to dehydrate (Bell et al. 2007).
The ootheca left behind by many species of cockroach is susceptible to parasitoids. Because of the size of E. floridana and the specialization parasitoids often exhibit towards specific hosts, Evaniidae, a common family of cockroach parasitoids, is ineffective against the Florida woods cockroach (Deyrup & Atkinson 1993). Among the Chalcidoidea, there is one species described in 1954 by Roth and Willis, Anastatus floridanus, as being a specialized parasitoid of E. floridana.
Feeding and Habitat (Back to Top)
The Florida woods cockroach feeds primarily on decomposing plant matter and is effectively a detritivore. Reasonable food sources could include dead or dying plant matter such as leaves and bark, as well as lichens, mosses, molds, soil microbes and mycoflora that would be encountered easily in damp dark environments (Bell et al. 2007). Because of these feeding habits, the species does not generally benefit from invading urban dwellings. However, these food sources may be present in nearby areas such as tree lines, decaying logs, under dense leaf litter, compost piles and other suitable locations.
This species tends to reside in stable sheltered areas. In populations outside of peridomestic scenarios, common habitations would include the cavities in trees or stumps, under tree bark, limestone cavities, leaf litter and densely shaded shrubs (Friauf 1953, Young and Cantrall 1958, Smith & Whitman 1992, Robinson 2005). In peridomestic environments, habitations can include holes in signs, around the base of ornamental trees such as coconut and palm, woodpiles and other shaded areas (Brenner and Pierce 1991, Young and Cantrall 1958, and Smith & Whitman 1992).
Defenses (Back to Top)
Many members of the genus Eurycotis generate defensive secretions as adults; in E. floridana the secretions are stored in a gland on the underside of the abdomen, oriented towards the cerci (Stay 1957). The chemicals are biologically created without dietary sequestration (Blum 1981, 1987). After discharge, it can take up to 30 days to replenish the contents of the gland (Farine et al. 2000), likely because of de novo chemical production and intense resource investment akin to venom production in some animals.
The chemicals stored by adult E. floridana function as an allomone, repelling antagonizers of the individual, as well as serving as an alarm pheromone to elicit an escape response in other members of the species (Farine et al. 1997). As a defensive chemical, the oily, odiferous secretion is harmful to skin and sensitive tissue such as the eyes. Direct contact is a potent irritant, with similar effects as defensive secretions used by coreids and pentatomids (Hemiptera), even being toxic to the producer (Turnbull & Fashing 2002, Robinson 2005). The chemical is strong enough to significantly irritate other species of cockroaches, ants, various invertebrate predators, and to deter small vertebrate predators such as lizards, birds, frogs and rodents (Eisner et al. 1959, O'Connell & Reagle 2002, Turnbull & Fashing 2002). As an alarm pheromone, low doses provoked strong escape reactions in nymphs and females, with reduced response in males (Farine et al. 1997).
This species can exhibit some directional control of the secretion and has variable spray distance, from as close as three centimeters to as distant as one meter (Robinson 2005). Early nymphal stages do not possess the ability to use the adult defensive secretions; the only known defense among nymphs is the ability to use defensive glues (Abed et al. 1994). Although the gland for the defensive secretion is present in the last nymphal stage, it does not produce the chemical until after molting into a reproductive adult (Stay 1957).
Management (Back to Top)
Florida Insect Management Guide for cockroaches
Selected References (Back to Top)
- Abed D, Farin JP, Francois J, Brossut R. 1994. Protein and amino acid composition of the tergal gland secretions of Blatta orientalis and Eurycotis floridana (Dictyoptera, Blattidae). Chemoecology 516: 19-25.
- Atkinson TH, Koehler PG, Patterson RS. 1990. Annotated checklist of the cockroaches of Florida (Dictyoptera: Blattaria: Blattidae, Polyphagidae, Blatellidae, Blaberidae). Florida Entomologist 73: 303-327.
- Barth RH JR. 1968. The mating behavior of Eurycotis floridana (Walker) (Blattaria, Blattoidea, Blattidae, Polyzosteriinae). Psyche (Cambridge) 75: 274-284.
- Barth RH JR. 1970. The mating behavior of Periplaneta americana (Linnaeus) and Blatta orientalis Linnaeus (Blattaria, Blattoidea, Blattidae, Blattinae) with notes on the mating behavior of three additional species of Periplaneta and on the interspecific effectiveness the female sex pheromones of cockroaches. Zeitschrift für Tierpsychologie 27: 722-748.
- Bell WJ, Louis MR, Nalepa CA. 2007. Cockroaches: Ecology, Behavior, and Natural History. pp. 116-130, 158-170. Johns Hopkins University Press, Baltimore, MD.
- Blum MS. 1981. Chemical Defenses of Arthropod Exocrine Compounds. pp. 40-562. Academic Press, New York, NY.
- Blum MS. 1987. Biosynthesis of arthropod exocrine compounds. Annual Review of Entomology 32: 381-413.
- Brenner RJ, Pierce RR. 1991. Seasonality of peridomestic cockroaches (Blattoidea, Blattidae) - Mobility, winter reduction, and effect of traps and baits. Journal of Economic Entomology 84: 1735-1745.
- Cornwell PB. 1968. The Cockroach: A Laboratory Insect and an Industrial Pest, Vol. 1. pp. 85-86. Hutchinson & Co LTD, London.
- Dakin ME, Hays KL. 1970. A synopsis of Orthoptera (sensu lato) of Alabama. Alabama Agricultural Experiment Station Bulletin 404: 118.
- Deyrup M, Atkinson TH. 1993. Survey of Evaniid wasps (Hymenoptera: Evaniidae) and their cockroach hosts (Blattodea) in a natural Florida habitat. Florida Entomologist 76: 589-593.
- Eisner T, Mckittrick F, Payne R. 1959. Defense sprays of roaches. Pest Control 27: 44-45
- Farine JP, Le Quere JL, Duffy J, Everaerts C, Brossut R. 1994. Male sex pheromone of cockroach Eurycotis floridana (Walker) (Blattidae, Polyzosteriinae): Role and composition of tergites 2 and 8 secretions. Journal of Chemical Ecology 20: 2291-2306.
- Farine JP, Everaerts C, Le Quere JL, Semon E, Henry R, Brossut R. 1997. The defensive secretion of Eurycotis floridana (Dictyoptera, Blattidae, Polyzosteriinae): Chemical identification and evidence of an alarm function. Insect Biochemistry and Molecular Biology 27: 577-586.
- Farine JP, Everaerts C, Abed D, Brossut R. 2000. Production, regeneration and biochemical precursors of the major components of the defensive secretion of Eurycotis floridana (Dictyoptera, Polyzosteriinae). Insect Biochemistry and Molecular Biology 30: 601-608.
- Friauf JJ. 1953. An ecological study of the Dermaptera and Orthoptera of the Welaka area in northern Florida. Ecological Monographs 23: 79-126.
- Gurney AB. 1959. New records of Orthoptera and Dermaptera from the United States. Florida Entomologist 42: 75-80.
- Hagenbuch BE, Koehler PG, Patterson RS, Brenner RJ. 1988. Peridomestic cockroachces (Orthoptera: Blattidae) of Florida: their species composition and suppression. Journal of Medical Entomology 25: 277-380.
- Hebard M. 1917. The Blattidae of North America North of the Mexican boundary. Memoirs of the American Entomological Society 2: 1-284.
- Mullen GR, Durden LA. 2009. Medical and Veterinary Entomology, 2nd edition: Cockroaches (Blattaria). pp. 46-47. Elsevier, Amsterdam.
- O'Connell TJ, Reagle NZ. 2002. Is the chemical defense of Eurycotis floridana a deterrent to small mammal predators? Florida Scientist 65: 245-249.
- Rehn JAG. 1902. Studies in American Blattidae. Transactions of the American Entomological Society 29: 259-290.
- Robinson WH. 2005. Urban Insects and Arachnids: A Handbook of Urban Entomology. pp. 52. Cambridge University Press, Cambridge, UK.
- Roth LM, Willis ER. 1954. Anastus floridanus (Hymenoptera: Eupelmidae) a new parasite on the eggs of the cockroach Eurycotis floridana. Transactions of the American Entomological Society 80: 29-41.
- Smith EH, Whitman RC. 1992. NPMA Field Guide to Structural Pests. pp. 4.8.1-4.8.2. National Pest Management Association.
- Stay B. 1957. The sternal gland of Eurycotis floridana (Blattaria, Blattidae). Annals of the Entomological Society of America 50: 514-519.
- Turnbull MW, Fashing NJ. 2002. Efficacy of the ventral abdominal secretion of the cockroach Eurycotis floridana (Blattaria: Blattidae) as a defense allomone. Journal of Insect Behavior 15: 369-384.
- Willis ER, Riser GR, Roth LM. 1958. Observations on reproduction and development in cockroaches. Annals of the Entomological Society of America 51: 53-69.
- Young FN, Cantrall IJ. 1958. Unheralded nobility: The cockroaches. The American Biology Teacher 20: 187-192.