common name: lesser mealworm, litter beetle
scientific name: Alphitobius diaperinus (Panzer) (Insecta: Coleoptera: Tenebrionidae)
Introduction - Synonymy - Distribution - Description - Life Cycle and Biology - Damage - Medical Importance - Management - Selected References
Introduction (Back to Top)
The lesser mealworm, Alphitobius diaperinus (Panzer), is a cosmopolitan general stored products pest of particular importance as a vector and competent reservoir of several poultry pathogens and parasites. It can also cause damage to poultry housing and is suspected to be a health risk to humans in close contact with larvae and adults. Adults can become a nuisance when they move en masse toward artificial lights generated by residences near fields where beetle-infested manure has been spread (Axtell 1999). Alphitobius diaperinus inhabits poultry droppings and litter and is considered a significant pest in the poultry industry.
Figure 1. Adult male lesser mealworm, Alphitobius diaperinus (Panzer). This specimen taken from Henderson County, North Carolina. Photograph by James Dunford, University of Florida.
Numerous studies have been conducted on lesser mealworm biology, physiology, and management. Lambkin (2001) conducted a thorough review of relevant scientific literature in reference to A. diaperinus and provides a good understanding of the biology, ecology and bionomics of the pest. Bruvo et al. (1995) conducted molecular work to determine satellite DNA variants on the chromosomes of A. diaperinus.
Alphitobius diaperinus is a member of the tenebrionid tribe Alphitobiini (Doyen 1989), which comprises four genera worldwide (Aalbu et al. 2002). Two genera occur in the United States, of which there are two species in the genus Alphitobius. There are approximately eleven known Alphitobius spp. worldwide, and additional species are encountered in stored products (Green 1980). The other known species in the United States, A. laevigatus (Fabricius) or black fungus beetle, is less commonly encountered and may also vector pathogens and parasites and occasionally cause damage to poultry housing.
Synonymy (Back to Top)
(from Aalbu et al. 2002)
Alphitobius Stephens (1832)
Heterophaga Redtenbacher (1845)
Cryptops Solier (1851)
(from Dunford 2000)
diaperinus (Panzer) (1797)
Tenebrio diaperinus Panzer (1797)
Phaleria diaperinus (Panzer), Latreille (1804)>Uloma opatroides Dejean (1821)
Uloma mauritanica Curtis (1831)
Alphitobius mauritanicus (Curtis), Stephens (1832)
Heterophaga opatroides (Dejean), Dejean (1833)
Heterophaga diaperina (Panzer), Redtenbacher (1849)
Alphitobius diaperinus (Panzer), Wollaston (1854)
Distribution (Back to Top)
Presently known worldwide, A. diaperinus is hypothesized to have originated in sub-Saharan Africa (Geden and Hogsette 1994, Lambkin 2001) and is considered a tropical exotic species in North America where it has been introduced from Europe.
In Florida, it likely occurs throughout the state; confirmed records include the following counties: Alachua, Broward, Charlotte, Clay, Dade, Hillsborough, Indian River, Manatee, Marion, Orange, Pasco, Pinellas, Polk, Putnam, and Volusia (Peck and Thomas 1998).
Additional confirmed records for the eastern United States (east of the Mississippi River) include the following states: North Carolina, West Virginia (Castrillo and Brooks 1998); Virginia (Geden and Steinkraus 2003); Georgia, Indiana, Michigan, New York, Ohio, and Wisconsin (Dunford and Young 2004, Dunford et al. 2005); Maine (Kaufman et al. 2005).
Description (Back to Top)
Adults: Lesser mealworm adults are broadly-oval, moderately convex, black or brownish-black and usually shiny in appearance. Color can be variable depending on age or 'strain' of lesser mealworm. Length is approximately 5.8 to 6.3 mm. Antennae are densely clothed with short yellowish hairs, with the terminal segment lighter in color. The head is deeply emarginate in front, has a distinct clypeal groove, and the surface is coarsely punctured. Eyes are also emarginated.
The pronotum is twice as broad as long, slightly narrowed from base to apex, with sides feebly curved and narrowly margined. The apex of the pronotum is broadly emarginated with apical angles prominent. The pronotal base is bisinuate with basal angles rectangular. There is a shallow impression on each side of the pronotum in the middle near the base. The pronotal disk is finely and sparsely punctured, with punctures much coarser laterally.
The elytra have moderately impressed striae with finely punctured, feebly convex intervals. Elytral punctures are sparse and nearly as large as those of the striae. The ventral surface of the lesser mealworm is dark reddish-brown, with the prosternal process horizontal between coxae and having a prominent apex.
Figure 2. Adult male lesser mealworm, Alphitobius diaperinus (Panzer). This specimen taken from University of Florida lab colony, (Denmark strain). Photograph by James Dunford, University of Florida.
Figure 3. Adult female lesser mealworm, Alphitobius diaperinus (Panzer). This specimen taken from University of Florida lab colony, (Denmark strain). Photograph by James Dunford, University of Florida.
Key to adult Alphtitobius Stephens species in the United States
1. Eyes deeply emarginate (completely or almost completely divided); prosternal process convex between forecoxae . . . . . laevigatus (Fabricius)
1'. Eyes emarginate (approximately one-half length of eye); prosternal process horizontal between forecoxae . . . . . diaperinus (Panzer)
coxa (pl. coxae): The basal segment of the leg.
prosternal process: A posterior prolongation of the prosternum behind forecoxae.
Figure 4. Ventral view of the head and forelegs of an adult lesser mealworm, Alphitobius diaperinus (Panzer). The prosternal process is indicated by the white arrow. Photograph by James Dunford, University of Florida.
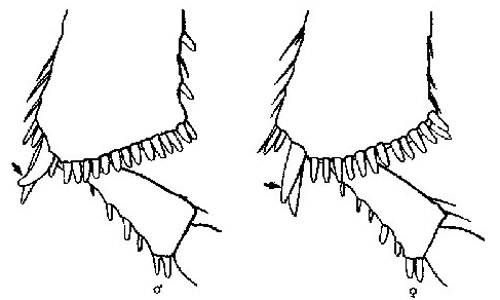
Figure 5. Spine on apex of median tibia (ventral side) showing difference between sexes (male-left, female-right) of Alphitobius diaperinus (Panzer) (from Bousquet 1990). Illustration by James Dunford, University of Florida.
Eggs: Eggs are approximately 1.5 mm in length, creamy white to tan colored, and slender with slightly rounded ends.
Larvae: Larvae have a segmented body with abdominal tip tapering posteriorly, three pairs of legs, and superficially resemble small wireworms or true mealworms (Tenebrio spp.). They are approximately 7 to 11 mm in length at last instar. Freshly-emerged larvae are a milky color. A shade of brown is observed after molting into the third instar to give a yellowish-brown appearance. The pale color tinge returns to that of the first/second instar larva when preparing to molt, however, it darkens again on the same day. There are approximately six to 11 larval instars.
Figure 6. Dorsal view (head on left) of larva of the lesser mealworm, Alphitobius diaperinus (Panzer). Photograph by James Dunford, University of Florida.
Figure 7. Ventral view (head on left) of larva of the lesser mealworm, Alphitobius diaperinus (Panzer). Photograph by James Dunford, University of Florida.
Figure 8. Lateral view (head on left) of larva of the lesser mealworm, Alphitobius diaperinus (Panzer). Photograph by James Dunford, University of Florida.
Pupae: Pupae are approximately 6 to 8 mm in length, creamy white to tan colored, with legs tucked alongside the body.
Figure 9. Pupa of the lesser mealworm, Alphitobius diaperinus (Panzer). Photograph by Jerry Butler and Jane Medley, University of Florida.
Life Cycle and Biology (Back to Top)
Alphitobius diaperinus is usually found infesting flour, meal, and other grain products, especially in poorly maintained grain processing plants (Spilman 1991). It has been associated with wheat, barley, rice, oatmeal, soybeans, cowpeas, and peanuts. It has also been reported from linseed, cottonseed, oilseed products, tobacco, skims, and drugs (Hosen et al. 2004). Because of its tropical origin, the lesser mealworm is well suited for warm, humid conditions. It is an important inhabitant of poultry or brooder houses where it lives in poultry droppings and litter. Both adults and larvae are abundant in manure from henneries and poultry farms (Francisco and Prado 2001).
This species has been observed in a wide variety of habitats such as bird nests, bat colonies/caves, and in rodent and other vertebrate living areas. Beetle populations in the hundreds of thousands have been found on and in caves inhabited by bats in various parts of the world, including Texas and Kenya (Falomo 1986). Larvae are known to feed on bat guano, mold, and on sick or dead bats, chickens, and pigeons (Falomo 1986). They also will feed on animal parts such as feathers, and other lesser mealworm individuals (cannibalism). Although Dermestes beetles are primarily known to clean carcasses for museum displays, the carnivorous habit of Alphitobius spp. is also used (Falomo 1986).
After mating, a female beetle has the potential to lay over 2,000 eggs (with an average closer to 200 to 400). Adults lay their eggs in cracks and crevices in the poultry house, in manure or litter, in grain hulls, and under feed and water lines. Adults can live three to twelve months, with females continuing to produce eggs most of their life at one to five day intervals.
Larvae hatch in four to seven days and complete development to the adult stage in 40 to 100 days, depending on temperature and food quality. Several studies have been conducted on A. diaperinus development on varied diets, at varying ambient temperatures and microhabitats, and among other poultry house inhabiting species (Calibeo-Hayes et al. 2005; Chernaki and de Almeida 2001; Hosen et al. 2004; Sarin and Saxena 1975; Strother and Steelman 2001; Renault et al. 2002, 2003a,b 2004; Watson et al. 2001). Larvae consume spilled feed, manure, and in some cases dead or sick birds and cracked eggs.
There are approximately six to 11 larval instars, and environmental conditions required for optimum development are 30° to 33°C with approximately 90% relative humidity. Francisco and Prado (2001) determined mean cephalic capsule sizes and developmental rate for larvae reared at 27°C. Mature larvae disperse, especially when crowded, to find isolated pupation sites, and often tunnel into thermal insulation materials where they construct pupal cells. The tunnels are expanded further when adult beetles eclose to leave the tunnels to find food. Both the larval and adult stages are primarily nocturnal, with greatest activity occurring at dusk. They are very active and quickly burrow into the litter when disturbed. Adults are long lived, normally persisting for more than a year, and under experimental conditions have survived for more than two years (Falomo 1986).
Damage (Back to Top)
Although A. diaperinus is not considered of major economic importance to whole grains, it does occur commonly on products already damaged by other biological agents, especially molds. However, it is of considerable importance in the poultry business as an avian disease vector, and there are human health risks associated with exposure to A. diaperinus. Poor community relations can also develop with, costly litigation when adults fly en masse to the artificial lights at private residences from beetle-infested manure (Hinchey 1997).
Lesser mealworms can also cause poultry house structural damage. When searching for suitable pupation sites, larvae will chew holes in styrofoam, fiberglass, and polystyrene insulation panels in the walls of poultry houses. The resulting damage can cause increased heating bills and additional building repair costs when the infested area is replaced. Energy costs in beetle-damaged broiler houses are reported to be 67% higher than in houses without beetle damage (Geden and Hogsette 1994).

Figure 10. Damage on wooden pillar caused by larvae of the lesser mealworm, Alphitobius diaperinus (Panzer). Photograph by Cornell University.
Watson et al. (2001) examined the interaction of the house fly, Musca domestica L., the filth fly predator Carcinops pumilio (Erichson), and A. diaperinus, and determined that A. diaperinus may have a negative impact on the establishment of C. pumilio in caged layer poultry houses. Lesser mealworm larvae can significantly reduce the survival of C. pumilio eggs and larvae, thus reducing their efficiency at suppressing M. domestica populations (Kaufman et al. 2002, Watson et al. 2001). However, lesser mealworm populations in high-rise caged layer houses may actually perform some beneficial functions. When beetle activity is intense, manure may be aerated and dried, and the beetles may become facultative predators of fly larvae (Geden and Hogsette 1994).
Figure 11. Adult lesser mealworms, Alphitobius diaperinus (Panzer), on poultry refuse. Photograph by Cornell University.
Figure 12. Adult lesser mealworms, Alphitobius diaperinus (Panzer), on poultry refuse. Photograph by Cornell University.
Figure 13. Adult lesser mealworms, Alphitobius diaperinus (Panzer), on poultry refuse. Photograph by Cornell University.
Medical Importance (Back to Top)
Lesser mealworms are important vectors of a number of poultry pathogens and parasites, such as the viruses of leucosis or Marek's disease, Gumboro disease (Falomo 1986), turkey coronavirus (Calibeo 2002, Watson et al. 2000), Newcastle disease, avian influenza (Hosen et al. 2004); bacteria such as Salmonella typhimurium (Loeffler) Escherichia coli (Migula), Aspergillus spp. and Staphylococcus ssp. (Chernaki-Leffer 2002, De Las Casas et al., 1968, Harein et al. 1970, McAllister et al. 1996,); protozoans such as Eimeria spp. that cause coccidiosis (Goodwin and Waltman 1996, Hosen et al. 2004); fungi (De Las Casas et al. 1972, Eugenio et al. 1970); helminths such as the nematode Subulura brumpti Lopes-Neira (Karunamoorthy et al. 1994); and fowl cestodes (Elowni & Elbihari 1979).
Infectious bursal disease (gumboro) in commercial broilers
Both adults and larvae of A. diaperinus can cause intestinal obstruction in poultry for slaughter since these birds lack chitinasis (enzyme to digest chitin) (Elowni & Elbihari 1979). This may eventually cause microscopic lesions along the bird's intestinal wall.
Another area of concern regarding A. diaperinus is associated health problems in humans. Tenebrionid beetles, including A. diaperinus, produce highly reactive benzoquinones as defense against predation (Tschinkel 1975). Quinones can be hazardous to human health and cause health risks when exposed to the insect for extended periods. Reported health related ailments caused by A. diaperinus include symptoms of asthma, headaches, dermatitis, allergic angioedema, rhinitis, erythema (reddening), and formation of papules (Falomo 1986, Schroeckenstein et al. 1988, Tseng et al. 1971). Exposure to quinone vapors can also result in conjunctivitis and corneal ulceration (Falomo 1986, Schroeckenstein et al. 1988).
It may be important for individuals with known insect allergies to avoid lesser mealworms as much as possible. Wearing protective gloves and masks while working under filtered and vented conditions may be required if conducting research on A. diaperinus. Quinones produced by tenebrionids are also suspected carcinogens; thus, quinone producing insects also represent a health hazard at all levels of food production and distribution (Ladisch 1965, Phillips and Burkholder 1984).
Management (Back to Top)
A variety of methodologies to help control the presence of A. diaperinus, and its impacts on poultry have been examined. Active beetle populations in manure or litter can be monitored by utilizing a variety of sampling methods, including Berlese funnels and tube traps (Safrit and Axtell 1984, Stafford et al. 1988). Buildings can also be surveyed by counting larvae on walls and posts in the early evening and "sentinel" insulation can be placed on walls and posts and monitored for entry holes on a regular basis (Geden and Hogsette 1994).
Identification and testing of aggregation and sex pheromones may lead to more effective monitoring methods and development of more effective baits. Pheromone releasing behaviors of A. diaperinus were examined by Falomo (1986). Geden and Hogsette (1994) recommend several basic and control component research needs for lesser mealworm control. A number of different control measures are warranted in a successful pest management approach.
Biological control: Several natural enemies of lesser mealworm have been found, but there are no current biological control options for A. diaperinus. The fungal pathogen Beauveria bassiana (Balsamo) Vuillemin holds the greatest promise for further development as a biological control agent. Geden et al. (1998) reported that larvae were highly susceptible to natural epizootic strains of B. bassiana isolated from A. diaperinus. Granular formations of B. bassiana strains provided 60 to 90% suppression in lesser mealworm larvae (Geden and Steinkraus 2003). Numerous studies on the genetic variation of B. bassiana and A. diaperinus populations have been conducted (Castrillo and Brooks 1998, Castrillo et al. 1999). Natural protozoan infections in A. diaperinus examined by Apuya et al. (1994) suggest that they may be an important, overlooked factor in regulating insect populations.
Steinernematid nematodes do not appear to provide long-term control or may be significantly effective in only one life stage (Alves et al. 2005, Geden et al. 1987, Szalanski et al. 2004), although additional testing of a number of different species and strains may be warranted.
The web contents of two spider species were found to have high numbers of A. diaperinus adults in Brazil (Rossi and Godoy 2005). Rossi and Godoy (2005) suggest that natural predation by spiders in poultry houses should be quantified in further studies for use in combination with other biological control agents. Geden and Hogsette (1994) recommend that additional surveys of beetle populations both in the United States and in the original home range of the lesser mealworm should be conducted to discover novel biological control agents.
Chemical control: Several formulations of carbaryl are registered for use against the lesser mealworm, including wettable powders, dusts, sprayable liquids, and baits. Several pyrethroids are registered and have been used as premise treatments (Geden and Hogsette 1994, Salin et al. 2003), and boric acid is available as a soil and premise treatment in some states (Geden and Hogsette 1994). Lime hydrate (calcium hydroxide) provided increased mortality among adult and larval A. diaperinus in the laboratory but has not been evaluated in the field (Watson et al. 2003).
None of the currently available insecticides provide satisfactory control when beetle populations are at outbreak levels. Some resistance to insecticides has also been observed (Lambkin 2005). Combined adulticidal (pyrethroid: cyfluthrin) and larvicidal (insect growth regulator: triflumuron) products showed some control but was dependent on building characteristics and management practices. Although lesser mealworms appear to be susceptible to many residual insecticides, the effectiveness of premise treatments can be limited by the accumulation of dust on treated surfaces (Despins et al. 1991) and manure levels in buildings.
Cultural and mechanical control: Utilized in conjunction with other control practices, another important method used to manage or suppress A. diaperinus outbreaks is some measure of cultural or mechanical control. Cold weather is a simple cultural control method for producers in northern locations, as most beetles can be eliminated by exposing them to sub-freezing temperatures for a week or more, as well as removing manure at these times. However, sub-freezing temperatures can damage pipes and kill birds.
Frequent manure cleanout or removal of litter and replacement of fresh shavings in poultry houses can help greatly reduce beetle populations (Geden and Hogsette 1994; Hinton and Moon 2003). Frequent removal of manure from caged layer systems also eliminates beetle problems. Application methodologies of manure in agricultural fields to suppress lesser mealworm populations were examined by Calibeo-Hayes et al. (2005) and Kaufman et al. (2005a).
Some types of insulation are more resistant to burrowing damage caused by the lesser mealworm and mechanical barriers can be used to prevent beetles from reaching susceptible building construction (Geden and Carlson 2001; Kaufman et al. 2005b).
Selected References (Back to Top)
- Aalbu RL, Triplehorn CA, Campbell JM, Brown KW, Somerby RA, Thomas DB. 2002. Family 106. Tenebrionidae Latreille 1802, p. 463-509. In: American Beetles. Volume 2. Polyphaga: Scarabaeoidea through Curculionoidea. (Arnett Jr RH, Thomas MC, Skelley PE, Frank JH, eds.). CRC Press, Boca Raton, FL.
- Alves LFA, Rohde C, VS Alves. 2005. Patogenicidade de Steinernema glaseri e S. carpocapsae (Nematoda: Rhabdita) contra o cascudinho, Alphitobius diaperinus (Panzer) (Coleoptera: Tenebrionidae). Neotropical Entomology 34: 139-141.
- Apuya LC, Stringham SM, Arends JJ, Brooks WM. 1994. Prevalence of protozoan infections in darkling beetles from poultry houses in North Carolina. Journal of Invertebrate Pathology 63: 25-259.
- Axtell RC. 1999. Poultry integrated pest management: status and future. Integrated Pest Management Reviews 4: 53-73.
- Bousquet Y. 1990. Beetles associated with stored products in Canada: An identification guide. Research Branch Agriculture Canada Publication 1837. Canadian Government Publishing Centre Supply and Services Ottawa, Canada. 214 pp.
- Bruvo B, Plohl M, Ugarkovic D. 1995. Uniform distribution of satellite DNA variants on the chromosome of Tenebrionid species Alphitobius diaperinus and Tenebrio molitor. Hereditas 123: 69-75.
- Buss LJ, Fasulo TR. (2006). Stored Product Pests. UF/IFAS. SW 185. CD-ROM.
- Butcher GD, Miles RD. (2003). Infectious bursal disease (gumboro) in commercial broilers. EDIS. http://edis.ifas.ufl.edu/VM024 (27 February 2006).
- Calibeo DR. 2002. Role and mitigation of two vectors of turkey coronavirus, Musca domestica L. and Alphitobius diaperinus (Panzer). Thesis, North Carolina State University.
- Calibeo-Hayes D, Denning SS, Stringham SM, Watson DW. 2005. Lesser mealworm (Coleoptera: Tenebrionidae) emergence after mechanical incorporation of poultry litter into field soils. Journal of Economic Entomology 98: 229-235.
- Castrillo LA, Brooks, WM. 1998. Differentiation of Beauveria bassiana isolates from the darkling beetle, Alphitobius diaperinus, using isozyme and RAPD analyses. Journal of Invertebrate Pathology 72: 190-196.
- Castrillo LA, Wiegmann BM, Brooks WM. 1999. Genetic variation in Beauveria bassiana populations associated with the darkling beetle, Alphitobius diaperinus. Journal of Invertebrate Pathology 73: 269-275.
- Chernaki-Leffer AM, de Almeida LM. 2001. Thermal requirements, development and survival of the immature stages of the Alphitobius diaperinus (Panz.) (Coleoptera: Tenebrionidae). Neotropical Entomology 30: 365-368.
- Chernaki-Leffer AM, Biesdorf SM, de Almeida LM, Leffer EVB, Vigne P. 2002. Isolation of enteric and litter organisms from Alphitobius diaperinus in brooder chicken houses in West of Parana State, Brazil. Revista Brasileira de Ciencia Avicola 4: 243-247.
- Curtis J. 1831. In: British Entomology, being illustrations and descriptions of the genera of insects found in Great Britain and Ireland: containing coloured figures from nature of the most rare and beautiful species, and in many instances of the plants upon which they are found. L Reeve and Company, London, 16 Volumes.
- Dejean PFMA. 1821. Catalogue de la collection de Coléoptères de M. le Baron Dejean. Paris, 136 pp.
- Dejean PFMA. 1833. Catalogue des Coléoptères de la collection de M. le Comte Dejean. Paris, 3: 177-256.
- De Las Casas E, Harein PK, Pomeroy BS. 1972. Bacteria and fungi within the mealworm collected from poultry brooder houses. Environmental Entomology 1: 27-30.
- De Las Casas E, Pomeroy BH, Harein PK. 1968. Infection and quantitative recovery of Salmonella typhimurium and Escherichia coli from within the lesser mealworn, Alphitobius diaperinus. Poultry Science 48: 1871-75.
- Despins JL, Turner Jr. EC, Pfeiffer DG. 1991. Evaluation of methods to protect poultry house insulation from infestations by lesser mealworm (Coleoptera: Tenebrionidae). Journal of Agricultural Entomology 8: 209-217.
- Doyen JT. 1989. Reconstitution of Coelometopini, Tenebrionini and related tribes of America north of Colombia (Coleoptera: Tenebrionidae). Journal of the New York Entomological Society 97: 277-304.
- Dunford JC. 2000. The darkling beetles of Wisconsin (Coleoptera: Tenebrionidae): taxonomy, natural history, and distributions. Thesis, University of Wisconsin-Madison. 291 pp.
- Dunford JC, Young DK. 2004. An annotated checklist of Wisconsin darkling beetles (Coleoptera: Tenebrionidae) with comparisons to the western Great Lakes fauna. Transactions of the American Entomological Society 130: 57-76.
- Dunford JC, Thomas MT, Choate Jr. PM. (2005). The darkling beetles of Florida and the eastern United States (Coleoptera: Tenebrionidae). http://entomology.ifas.ufl.edu/teneb/index.htm (7 January 2006)
- Elowni EE, Elbihari S. 1979. Natural and experimental infection of the beetle, Alphitobius diaperinus (Coleoptera: Tenebrionidae) with Choanotaenia infundibulum and other chicken tapeworms. Veterinary Science Communications 3: 171-173.
- Eugenio C, De Las Casas E, Harein PK, Mirochia CJ. 1970. Detection of the mycotoxin F-2 in the confused flour beetle and lesser mealworm. Journal of Economic Entomology 63: 412-415.
- Falomo AA. 1986. The Pheromone Biology of the Lesser Mealworm Alphitobius diaperinus (Panzer), (Coleoptera: Tenebrionidae). Thesis, University of Wisconsin-Madison.
- Francisco O, Prado, AP do. 2001. Characterization of the larval stages of Alphitobius diaperinus (Panzer) (Coleoptera: Tenebrionidae) using head capsule width. Revista Brasileira de Biologia 61: 125-131.
- Geden CJ, Arends JJ, Axtell RC. 1987. Field trials of Steinernema feltiae (Nematoda: Steinernematidiae) for control of Alphitobius diaperinus (Coleoptera: Tenebrionidae) in commercial broiler and turkey houses. Journal of Economic Entomology 80: 136-141.
- Geden CJ, Arends JJ, Rutz DA, Steinkraus DC. 1998. Laboratory evaluation of Beauveria bassiana (Moniliales: Moniliaceae) against the lesser mealworm, Alphitobius diaperinus (Coleoptera: Tenebrionidae), in poultry litter, soil, and a pupal trap. Biological Control 13: 71-77.
- Geden CJ, Carlson DA. 2001. Mechanical barrier for preventing climbing by lesser mealworm (Coleoptera: Tenebrionidae) and hide beetle (Coleoptera: Dermestidae) larvae in poultry houses. Journal of Economic Entomology 94: 1610-1616.
- Geden CJ, Hogsette JA. (2001). Research and extension needs for integrated pest management for arthropods of veterinary importance. Center for Medical, Agricultural, and Veterinary Entomology USDA-ARS Workshop Proceedings, Lincoln, Nebraska. http://www.ars.usda.gov/Services/docs.htm?docid=10139 (21 March 2006).
- Geden CJ, Steinkraus DC. 2003. Evaluation of three formulations of Beauveria bassiana for control of lesser mealworm and hide beetle in Georgia poultry houses. Journal of Economic Entomology 96: 1602-1607.
- Goodwin MA, Waltman WD. 1996. Transmission of Eimeria, viruses, and bacteria to chicks: Darkling beetles (Alphitobius diaperinus) as vectors of pathogens. Journal of Applied Poultry Research 5: 51.
- Green M. 1980. Alphitobius viator Mulsant & Godart in stored products and its identification (Coleoptera: Tenebrionidae). Journal of Stored Products Research l6: 67-70.
- Harein PK, De Las Casas E, Pomeroy BS, York MD. 1970. Salmonella spp. and serotypes of Escherichia coli isolated form the lesser mealworm collected in poultry brooder houses. Journal of Economic Entomology 63: 80-81.
- Hinchey F. 1997. Bugged residents sue over beetles. The Columbus Dispatch. October 2, 1997.
- Hinton JL, Moon RD. 2003. Arthropod populations in high-rise, caged layer houses after three manure cleanout treatments. Journal of Economic Entomology 96: 1352-1361.
- Hosen M, Khan AR, Hossain M. 2004. Growth and development of the lesser mealworm, Alphitobius diaperinus (Panzer) (Coleoptera: Tenebrionidae) on cereal flours. Pakistan Journal of Biological Sciences 7: 1505-1508.
- Karunamoorthy G, Chellappa DJ, Anandari R. 1994. The life history of Subulura brumpti in the beetle Alphitobius diaperinus. Indian Veterinary Journal 71: 12-15.
- Kaufman PE, Burgess M, Rutz DA. 2002. Population dynamics of manure inhabiting arthropods under an integrated pest management (IPM) program in New York poultry facilities-3 case studies. Journal of Applied Poultry Research 11: 90-103.
- Kaufman PE, Reasor C, Murray KD, Waldron JK, Rutz DA. 2005b. Evaluation of a barrier to inhibit lesser mealworm (Coleoptera: Tenebrionidae) and Dermestidae movement in high-rise, caged-layer poultry facilities. Journal of Economic Entomology 98:1744-1749.
- Kaufman PE, Reasor C, Waldron JK, Rutz DA. 2005a. Suppression of adult lesser mealworm (Coleoptera: Tenebrionidae) using soil incorporation of poultry manure. Journal of Economic Entomology 98: 1739-1743.
- Ladisch RK. 1965. Quinone toxins and allied synthetics in carcinogenesis. Proceedings of the Pennsylvania Academy of Science 38: 144-149.
- Lambkin TA. 2001. Investigations into the management of the darkling beetle. Rural Industries Research and Development Corporation, Kingston, Australia. 99 pp.
- Lambkin TA. 2005. Baseline responses of adult Alphitobius diaperinus (Panzer) (Coleoptera: Tenebrionidae) to fenitrothion, and susceptibility status of populations in Queensland and New South Wales, Australia. Journal of Economic Entomology 98: 938-942.
- Latreille PA. 1804. Histoire Naturelle, Générale et Particulière des Crustacés et des Insectes. Paris, 10: 415 pp.
- McAllister JC, Steelman CD, Skeeles JK, Newberry LA, Gbur EE. 1996. Reservoir competence of Alphitobius diaperinus (Coleoptera: Tenebrionidae) for Escherichia coli (Eubacteriales: Enterobacteriaceae). Journal of Medical Entomology 33: 983-987.
- Panzer GWF. 1797. Faunae Insectorum Germanicae initia oder Deutschlands Insecten. Nürnberg. Volume 37: 1-60.
- Peck SB, Thomas MC. 1998. A Distributional Checklist of the Beetles (Coleoptera) of Florida. Arthropods of Florida and Neighboring Land Areas Department of Agriculture and Consumer Services, Gainesville, Florida 16: 1-166.
- Phillips JK, Burkholder WE. 1984. Health hazards of insects and mites in food. In Insect Management for Food Storage and Processing. (Bauer FJ, ed.). pp. 280–293. American Association of Cereal Chemists, St. Paul, MN.
- Redtenbacher W. 1845. Die Gattungen der deutschen Käfer Fauna nach der analytischen Methode Bearbeitet, nebst einem kurz gefassten Leitfaden, zum Studium dieses Zweiges der Entomologie. Wien 177 pp.
- Redtenbacher W. 1849. Fauna Austriaca. Die Käfer, nach der analytischen Methode Bearbeitet. Wien, 883 pp.
- Renault D, Hance T, Vannier G, Vernon P. 2003a. Is body size an influential parameter in determining the duration of survival at low temperatures in Alphitobius diaperinus Panzer (Coleoptera: Tenebrionidae)? Journal of Zoology 259: 381-388.
- Renault D, Hervant F, Vernon P. 2002. Comparative study of the metabolic responses during food shortage and subsequent recovery at different temperatures in the adult lesser mealworm, Alphitobius diaperinus (Coleoptera: Tenebrionidae). Physiological Entomology 27: 291-301.
- Renault D, Hervant F, Vernon P. 2003b. Effect of food shortage and temperature on oxygen consumption in the lesser mealworm, Alphitobius diaperinus (Panzer) (Coleoptera: Tenebrionidae). Physiological Entomology 28: 261-267.
- Renault D, Nedved O, Hervant F, Vernon P. 2004. The importance of fluctuating thermal regimes for repairing chill injuries in the tropical beetle Alphitobius diaperinus (Coleoptera: Tenebrionidae) during exposure to low temperatures. Physiological Entomology 29: 139-145.
- Rossi MN, Godoy WAC. 2005. Web contents of Nesticodes rufipes and Latrodectus geometricus (Araneaea: Theridiidae) in a Brazilian poultry house. Journal of Entomological Science 40: 347-351.
- Safrit RD, Axtell RC. 1984. Evaluations of sampling methods for darkling beetles (Alphitobius diaperinus) in litter of turkey and broiler houses. Poultry Science 63: 2368-2375.
- Salin C, Delettre YR, Vernon P. 2003. Controlling the mealworm Alphitobius diaperinus (Coleoptera: Tenebrionidae) in broiler and turkey houses: Field trials with combined insecticide treatment: Insect growth regulator and pyrethroid. Journal of Economic Entomology 96: 126-130.
- Sarin K, Saxena SC. 1975. Food preference and site of damage to preferred products by Alphitobius diaperinus (Panz.). Bulletin of Grain Technology 13:50-51.
- Schroeckenstein DC, Meier-Davis S, Graziano FM, Falomo A, Bush RK. 1988. Occupational sensitivity to Alphitobius diaperinus (Panzer) (lesser mealworm). Journal of Allergy and Clinical Immunology 82: 1081-1088.
- Solier AJJ. 1851. Orden III. Coleópteros, Pentamera et Heteromera. In Gay C, Historia fisica y politica de Chile. Zoologia, Volume 5, [Privately published], Paris, 285 pp.
- Spilman TJ. 1991. 11, Darkling Beetles (Tenebrionidae, Coleoptera). In Insect and Mite Pests in Food. (Gorham JR, ed). United States Department of Agriculture, Agricultural Handbook 655: 185-214, 589-598.
- Stafford KC, Collison CH, Burg JG, Cloud JA. 1988. Distribution and monitoring lesser mealworms, hide beetles, and other fauna in high-rise caged-layer poultry houses. Journal of Agricultural Entomology 5: 89-101.
- Stephens JF. 1832. Illustrations of British Entomology. Mandibulata. Baldwin and Cradock, London 5: 1-447.
- Strother KO, Steelman CD. 2001. Spatial analysis of Alphitobius diaperinus (Coleoptera: Tenebrionidae) in broiler production facilities. Environmental Entomology 30: 556-561.
- Szalanski AL, Palmer TW, McKay T, Steelman CD. 2004. Infectivity of Steinernema spp. (Nematoda: Steinernemtidae) to adult litter beetles, Alphitobius diaperinus (Coleoptera: Tenebrionidae) in the laboratory. Biocontrol Science and Technology 14: 81-85.
- Tschinkel, W.R. 1975. A comparative study of the chemical defensive system of tenebrionid beetles: chemistry of the secretions. Journal of Insect Physiology 21: 753-783.
- Tseng YL, Davidson JA, Menzer RE. 1971. Morphology and chemistry of the odoriferous gland of the lesser mealworm, Alphitobius diaperinus (Coleoptera: Tenebrionidae). Annals of the Entomological Society of America 64: 425-430.
- Watson DW, Denning SS, Zurek L, Stringham SM, Elliott J. 2003. Effects of lime hydrate on the growth and development of darkling beetle, Alphitobius diaperinus. International Journal of Poultry Science 2: 91-96.
- Watson DW, Kaufman PE, Rutz DA, Glenister CS. 2001. Impact of the darkling beetle Alphitobius diaperinus (Panzer) on establishment of the predaceous Carcinops pumilio (Erichson) for Musca domestica control in caged-layer poultry houses. Biological Control 20: 8-15.
- Watson DW, Guy JS, Stringham SM. 2000. Limited transmission of turkey coronavirus (TCV) in young turkeys by adult darkling beetles, Alphitobius diaperinus Panzer (Tenebrionidae). Journal of Medical Entomology 37:480-483.
- Wollaston TV. 1854. Insecta Maderensia; being an account of the insects of the islands of the Maderian group. London, 634 pp.